Animal ethics compliance software
Keep ethics approvals, procedures, people, and reporting connected.
AMS gives research institutions one place to manage animal ethics records, approval scope, certified users, SOPs, procedure usage, revisions, and reporting obligations.
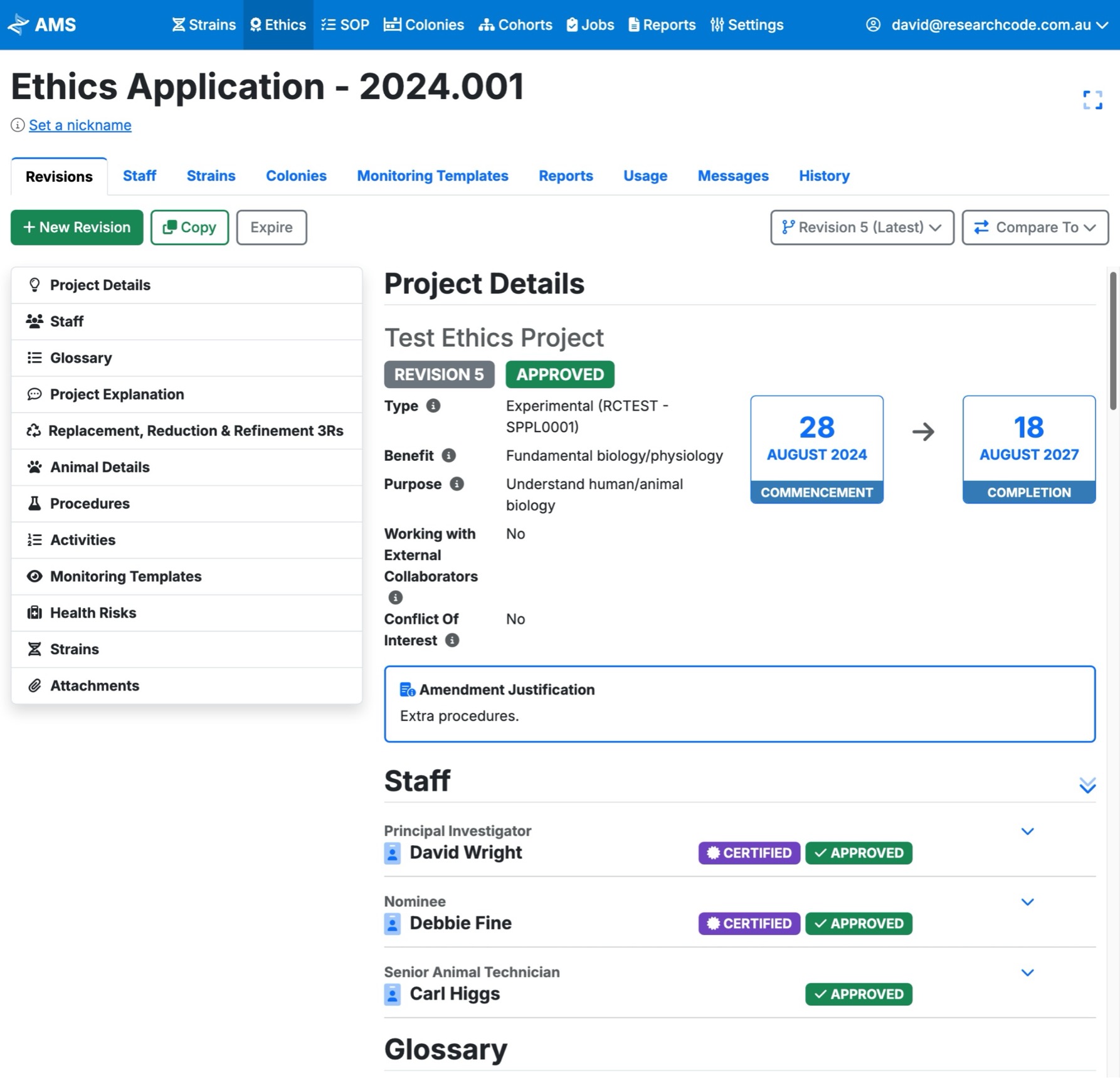
Approval scope in context
Connect each ethics application to approved species, strains, colonies, procedures, SOPs, staff, dates, and reporting requirements.
Checks where work happens
Surface approval and training context when procedures are planned or recorded, so compliance is part of daily work rather than a separate spreadsheet.
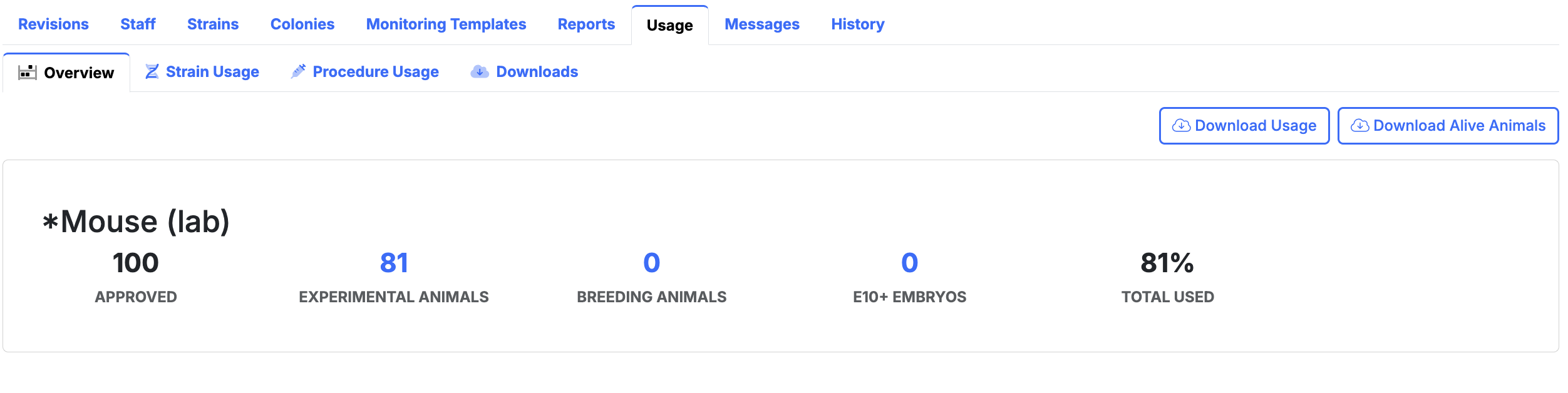
Records ready for review
Keep usage logs, revisions, attachments, and report history together for audits, committee review, annual reporting, and final reporting.
Built for Australian animal research governance
AMS has been developed with more than 20 years of experience in animal research systems and has been used in leading Australian research environments since 2016.
- Applications and revisions - retain version history and review context as projects change.
- Approved procedures - link methods, SOPs, users, species, strains, and colonies to the approval.
- Certified staff - connect training and procedure access to the people performing the work.
- Usage and animal use returns - report from actual recorded colony and procedure activity.
- Attachments and links - keep supporting documents close to the record they explain.
Who this helps
AMS supports the people who need ethics information to be accurate, current, and visible without adding manual administration.
Ethics officers and AEC teams
Review approval scope, revisions, reporting status, and usage history from one system of record.
Facility managers
Understand which work is approved, which staff are certified, and where compliance risk may emerge.
Researchers and project teams
Work within approved procedures and keep required records without duplicating information across systems.
Research operations and reporting teams
Prepare annual reports, final reports, animal use returns, and operational summaries from connected data.
See how AMS handles animal ethics compliance.
Book a walkthrough focused on your ethics approval, usage, and reporting workflows.
Request a Demo